STANDARD Q COVID-19 IgM/IgG Combo
This kit is a rapid immunochromatography test designed for the qualitative presumptive detection of specific IgM and IgG to 2019 novel coronavirus (nCoV) in humoral fluid.
STANDARD Q COVID-19 IgM/IgG Combo Test Kit
STANDARD Q COVID-19 IgM/IgG Combo Test Kit is a rapid immunochromatography test designed for the qualitative presumptive detection of specific IgM and IgG to SARS-CoV-2 in humoral fluid.
- Rapid testing for SARS-CoV-2 antibodies within 10~15 minutes
- Specimen : Whole blood (20ul), serum , plasma (10ul)
- Suitable for Point of Care Testing. No need for extra equipment
Email Us: medicalequip@indofuji.com
Information
Specification
| Information | Detail |
|---|---|
| Test time | Within 10~15 mins |
| Specimen | Whole blood/Serum/plasma |
| Storage temperature | 2-30℃/36-86℉ |
Ordering Information
| Cat. No. | Product | Storage temperature | Pack size |
|---|---|---|---|
| 09COV50G | STANDARD Q COVID-19 IgM/IgG Combo Test | 2-30℃/36-86℉ | 40Tests/kit |
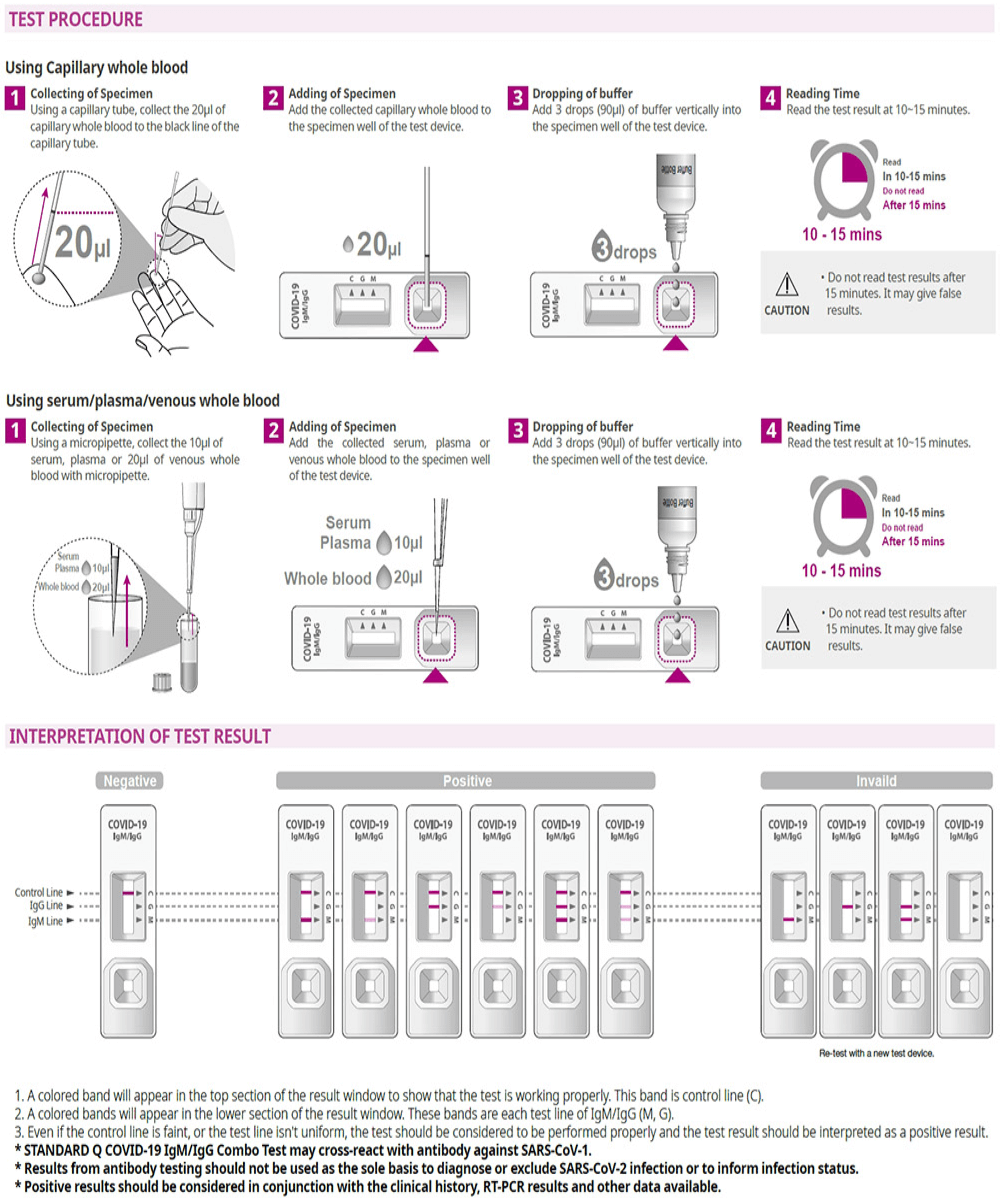
Instructions for Use
Performance Characteristics
[Clinical evaluation]
Performance characteristic for the STANDARD Q COVID-19 IgM/IgG Combo Test for rapid detection of anti-SARS-CoV-2 antibodies was established in retrospective, multi institutes, randomized, single-blinded study conducted at a trial site in KOREA during the 2020 SARS-CoV-2 pandemic situation. A total of 441 retrospective specimens were tested using the STANDARD Q COVID-19 IgM/IgG Combo Test. These specimens consisted of serum from PCR positive or negative confirmed patients. The performance of the STANDARD Q COVID-19 IgM/IgG Combo Test were compared to a commercialized molecular assay. Although the STANDARD Q COVID-19 IgM/IgG Combo Test allows to test for IgM and IgG separately, due to the differing inter-patient time response to the virus, any individual with positive result for the IgM or the IgG test should be read as a positive for anti-SARS-CoV-2 antibodies. The combined test result (positive for IgM and/or IgG or negative for IgM and/or IgG) was used to calculate the total test sensitivity and specificity.
Test sensitivity
The seroconversion time of IgM and IgG antibodies varies from person to person, but it was estimated to be around 7 days after onset of symptom4,5. The STANDARD Q COVID-19 IgM/IgG Combo Test showed 94.51% of sensitivity using specimens from patients 7 days after symptom onset (combined IgM+IgG).
Table 1. Summary of sensitivity of the STANDARD Q COVID-19 IgM/IgG Combo Test Compare to PCR confirmed specimens from 7 days after symptoms onset is 94.51%.
| ≥ 7 Days after symptom onset | PCR | |||
| Positive | Negative | Total | ||
STANDARD Q COVID-19 IgM/IgG Combo Test |
Positive | 155 | 0 | 155 |
| Negative | 9 | 0 | 9 | |
| Total | 164 | 0 | 164 | |
Sensitivity |
94.51%
|
|||
Table 2. Summary of sensitivity of the STANDARD Q COVID-19 IgM/IgG Combo Test compared to PCR confirmed specimens within less than 7 days after symptom onset is 69.05%.
| > 7 Days after symptom onset | PCR | |||
| Positive | Negative | Total | ||
STANDARD Q COVID-19 IgM/IgG Combo Test |
Positive | 29 | 0 | 29 |
| Negative | 13 | 0 | 13 | |
| Total | 42 | 0 | 42 | |
Sensitivity |
69.05%
|
|||
Table 3. Summary of sensitivity of the STANDARD Q COVID-19 IgM/IgG Combo Test compared to PCR confirmed specimens between 7 days to 14 days after symptom onset is 89.39%
| 7-14 Days after symptom onset | PCR | |||
| Positive | Negative | Total | ||
STANDARD Q COVID-19 IgM/IgG Combo Test |
Positive | 59 | 0 | 59 |
| Negative | 7 | 0 | 7 | |
| Total | 66 | 0 | 66 | |
Sensitivity |
89.39%
|
|||
Table 4. Summary of sensitivity of the STANDARD Q COVID-19 IgM/IgG Combo Test compared to PCR confirmed specimens from 14 days after symptom onset is 96.94%
| > 14 Days after symptom onset | PCR | |||
| Positive | Negative | Total | ||
STANDARD Q COVID-19 IgM/IgG Combo Test |
Positive | 95 | 0 | 95 |
| Negative | 3 | 0 | 3 | |
| Total | 98 | 0 | 98 | |
Sensitivity |
96.94%
|
|||
Test specificity
The STANDARD Q COVID-19 IgM/1gG Combo Test showed 95.74% of specificity (combined IgM+IgG).
Table 5. Summary of specificity of the STANDARD Q COVID-19 IgM/IgG Combo Test compared to PCR confirmed specimens is 95.74%
| PCR | ||||
| Positive | Negative | Total | ||
STANDARD Q COVID-19 IgM/IgG Combo Test |
Positive | 0 | 10 | 10 |
| Negative | 0 | 225 | 225 | |
| Total | 0 | 235 | 235 | |
Sensitivity |
95.74%
|
|||
Analytical Performance
- Limit of Detection: IgM-9.37 ug/ml, IgG-3.75 ug/ml
- Cross-Reactivity: No cross-reactivity for the specimen of HIV Seroconversion panel, Japanese Encephalitis positive, Zika virus positive, Chikungunya positive, Dengue IgM positive, Salmonella typhi IgM positive, Rubella IgM, CMV IgG/IgM, Tick borne encephalitis IgM positive, West Nile Virus positive, Treponema palladium, HAV IgM positive, HAV IgG positive, HBV Ab positive, HCV Ab positive, Influenza vaccine positive, Leishmania positive, Brucella IgM positive, Chagas positive, Toxoplasma positive, Filariasis positive, Mycoplasma pneumonia IgM positive, Mycoplasma pneumonia IgG positive, Mycoplasma pneumonia IgM positive, Influenza A IgM positive, Influenza B IgM positive, Influenza A and B IgG+IgM positive and Tuberculosis positive.
- Interference study: For Exogenous substances, there was no interference with medicines such as Zanamivir (for Influenza), Oseltamivir (for Influenza), Artemether-lumefantrine (for Malaria), Doxycycline hyclate (for Malaria), Quinine (for Malaria), Lamivudine (Retroviral medication), Ribavirin (for HCV), Daclatasvir (for HCV). Also there was no interference with Anti-inflammatory medication (Acetaminophen, Acetylsalicylic acid, Ibuprofen), Antibiotic (Erythromycin, Ciprofloxacin), Dietary substances (Caffeine, Ethanol, Biotin), Triglycerides, Cholesterol, Bilirubin(Unconjugate), Hemoglobin and Anticoagulants (EDTA, Heparin, Sodium citrate). For Edogenous substances, there was no interference with human anti-mouse antibody, whole blood of pregnant women and whole blood having elevated levels of hemoglobin, elevated levels of C-reactive protein and Elevated IgG or IgM samples.
- Matrix Equivalency: The matrix and anticoagulant does not affect the detection of COVID-19 IgG and IgM in contrived specimens between serum, plasma(Heparin, EDTA, Sodium citrate), venous whole blood(Heparin, EDTA, Sodium citrate), and capillary whole blood(collected in EDTA-treated tubes).
Limitation of Test
- The test procedure, precautions and interpretation of results for this test must be followed strictly when testing.
- This test detects the presence of SARS-CoV-2 IgM/IgG in the specimen and should not be used as the sole criteria for the diagnosis of SARS-CoV-2 infection.
- Test results must be considered with other clinical data available to the physician.
- For more accuracy of immune status, additional follow-up testing using other laboratory methods is recommended.
- Neither the quantitative value nor the rate anti- SARS-CoV-2 IgM/IgG concentration can be determined by this qualitative test.
- Failure to follow the test procedure and interpretation of test results may adversely affect test performance and/or produce invalid results.
- A negative result may occur if the concentration of antigen or antibody in a specimen is below the detection limit of the test or if the specimen was collected or transported improperly, therefore a negative test result does not eliminate the possibility of SARS-CoV-2 infection, and should be confirmed by viral culture or an molecular assay or ELISA.
- Positive test results do not rule out co-infections with other pathogens.
- Negative test results are not intended to rule in other coronavirus infection except the SARS-CoV-1.
- Children tend to shed virus for longer periods of time than adults, which may result in differences in sensitivity between adults and children.
Notification for COVID-19 Antibody Tests
- This test has not been reviewed by the FDA.
- Negative results do not rule out SARS-CoV-2 infection, particularly in those who have been in contact with the virus. Follow-up testing with a molecular diagnostic should be considered to rule out infection in these individuals.
- Results from antibody testing should not be used as the sole basis to diagnose or exclude SARS-CoV-2 infection or to inform infection status.
- Positive results may be due to past or present infection with non-SARS-CoV-2 coronavirus strains, such as coronavirus HKUI, NL63, OC43, or 229E or past or present infection with SARS virus (no. 6)
- Not for the screening of donated blood.
- The test procedure should be conducted in ambient temperature and pressure.
- Results of these tests should be appropriately recorded in a test report.